Now that you know about how blood works and the important role of hemoglobin, I’d like to explore the topic of sickle cell anemia as promised.
In the biology course Organisms and Environments we learned that the malaria virus is carried by a protist parasite called Plasmodium, which enters the human body via a mosquito vector. Malaria is the end product of a parasite chain, with mosquitoes carrying Plasmodium, which creates the disease symptoms.
 In Ecology and Population Biology, we learned that where malaria is present there is a higher prevalence of sickle cell anemia. Sickle cell anemia is almost non-existent in countries where malaria is not found, but something about the sickle cell disease produces resistance to malaria. In Genetics we learned that sickle cell anemia is a genetic disorder in which a change of one single nucleotide led to a novel amino acid substitution in the protein structure of hemoglobin. The disorder is autosomal recessive, so those that have two copies of the allele are fully affected, those that are heterozygous (two different copies) are only partially affected, and those that are homozygous dominant and don’t carry the mutated allele are not affected at all.
In Ecology and Population Biology, we learned that where malaria is present there is a higher prevalence of sickle cell anemia. Sickle cell anemia is almost non-existent in countries where malaria is not found, but something about the sickle cell disease produces resistance to malaria. In Genetics we learned that sickle cell anemia is a genetic disorder in which a change of one single nucleotide led to a novel amino acid substitution in the protein structure of hemoglobin. The disorder is autosomal recessive, so those that have two copies of the allele are fully affected, those that are heterozygous (two different copies) are only partially affected, and those that are homozygous dominant and don’t carry the mutated allele are not affected at all.Now, after all the build up between three of my biology courses, after years of asking “but how does sickle cell anemia produce resistance to malaria?” my question has finally been answered in Bio-Chemistry this year!
That single nucleotide change that is responsible for sickle cell anemia results in the inappropriate placement of a valine amino acid where a glutamate amino acid is supposed to be in the hemoglobin protein. Due to its polar qualities, the glutamate amino acid has no problem sitting on the outside of the hemoglobin protein facing the aqueous environment of the cell, and is therefore called “hydrophilic.” Valine, on the other hand, is a non-polar, uncharged amino acid and is said to be “hydrophobic.” Water tends to exclude this amino acid, pushing it away.
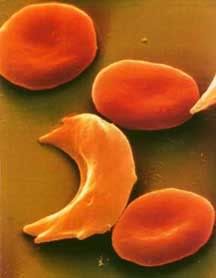 Hemoglobin in its relaxed state favors the binding of oxygen and has no hydrophobic sites revealed. When hemoglobin drops the oxygen and is in the tensed, deoxygenated state though, a hydrophobic niche is revealed, the perfect hiding place for that hydrophobic valine. If, by chance, the valine comes into contact with this site, it will not leave. Water excludes the hydrophobic molecules, forcing them to stick together. If enough hemoglobin valines find their way into this hydrophobic niche, aggregates of hemoglobin form in the erythrocyte (red blood cell), creating long hemoglobin strands. The cell will begin to form a sickle shape as a result of these strands and has potential to clot capillaries and even arteries if enough sickle cells are present. This is why it’s so dangerous for someone who has the sickle cell mutation to be in low oxygen environments; the less oxygen they are getting the higher the chance their hemoglobin will deoxygenate and aggregate to form sickle cells. This risk is so high in individuals who are homozygous for the mutation that they often die in childhood. People who are heterozygous for the mutation are somewhat better off. It’s still a risky disease, but their immune system does a pretty good job cleaning up those sickle shaped erythrocytes before they form clots.
Hemoglobin in its relaxed state favors the binding of oxygen and has no hydrophobic sites revealed. When hemoglobin drops the oxygen and is in the tensed, deoxygenated state though, a hydrophobic niche is revealed, the perfect hiding place for that hydrophobic valine. If, by chance, the valine comes into contact with this site, it will not leave. Water excludes the hydrophobic molecules, forcing them to stick together. If enough hemoglobin valines find their way into this hydrophobic niche, aggregates of hemoglobin form in the erythrocyte (red blood cell), creating long hemoglobin strands. The cell will begin to form a sickle shape as a result of these strands and has potential to clot capillaries and even arteries if enough sickle cells are present. This is why it’s so dangerous for someone who has the sickle cell mutation to be in low oxygen environments; the less oxygen they are getting the higher the chance their hemoglobin will deoxygenate and aggregate to form sickle cells. This risk is so high in individuals who are homozygous for the mutation that they often die in childhood. People who are heterozygous for the mutation are somewhat better off. It’s still a risky disease, but their immune system does a pretty good job cleaning up those sickle shaped erythrocytes before they form clots.Now, what does this have to do with malaria, you ask? Well, recall from the last article that there are a number of environmental signals that will cause hemoglobin to drop its oxygen. One of those signals is a low pH (acidity), as active tissues are often more acidic. When the parasite Plasmodium enters an erythrocyte the pH drops. The hemoglobin then drop their oxygen all at once, exposing the hydrophobic sites to the hydrophobic valines. They aggregate and cause the cell to sickle up, marking the cell for clean up by the immune system. The immune system is then able to sweep the parasite out, before it has a chance to reproduce!
Sources:
All info is from my coursework at UI
Images:
Sickle cells: http://www.defiers.com/scd.html
Plasmodium: wikipedia.com


0 Comments:
Post a Comment
<< Home