From Large to Small, Nothing Stands Still for Long...

One of the things that first fascinated me about biology is the general bustling nature of life. Organisms are constantly working, eating, breathing, and drinking to maintain the essential aspects of their complex internal environments. It’s a wonder that creatures can do it all--from the tiniest aphid to an enormous elephant, every organism must keep a steady temperature, protect against degradation, maintain bodily structure, and supply energy to its cells. The inside of a gnat has more “workers” than any factory and is more intricate and synchronized than any great city of the world.
Even the smallest levels of the orangism are busy at work. As I entered into cellular level biology, I realized the workings of any given cell alone are comparable to a small country. When we first lean the components of a cell in high school, we see it as this mainly empty compartment with a few organelles here and there. It is not until later that they tell us the cell is literally packed. Filled with water, we can envision it more as a sea of enzymes working away on their given tasks, filaments webbing the inside like a great monorail network, RNAs flying about like blueprints on the run, small molecules acting as precursors to great projects or messengers with the latest news, organelles operating like great factories spilling out their products, pores regulating the influx and efflux of this carefully constructed environment. Here is an image of epithelial (skin) cells where the microfilaments have been stained in red.
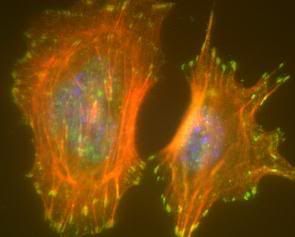
Although there are more components that are not shown, it is easy to see the cell is far from empty. To see other cytoskeleton structures that can exist in a cell look here.
Just to blow my mind a little further, my bio-chemistry instructor today mentioned “breathing proteins.” A weird phrase to hear at 7:30 in the morning, he had to repeate it once more for the confused and sleepy class. Proteins are usually thought of as strong, inflexible stuff, the scaffolding of bodily structures. At certain low temperatures, we can view proteins and discover their (nearly) immobile structures, but in the high heat liquid environment of the body, these molecules are allowed quite a bit of wiggle room. Each amino acid component of the protein has a degree of freedom to rotate, and all together they can scoot around and create variations in the overall shape of the macromolecule. Dr. Cole calls this “breathing.”
 Going deeper, we not only see that molecules like amino acids are active, but the very atoms and the bonds between them are as well. In the study of resonance structures in organic chemistry, we are shown that electrons are quite the flighty bunch. Take a look at this sulfate molecule below (also shown in 3D at left):
Going deeper, we not only see that molecules like amino acids are active, but the very atoms and the bonds between them are as well. In the study of resonance structures in organic chemistry, we are shown that electrons are quite the flighty bunch. Take a look at this sulfate molecule below (also shown in 3D at left):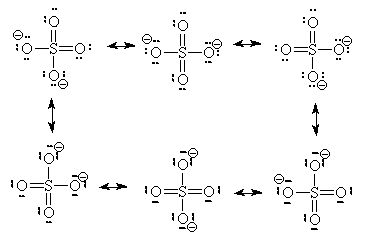
As you can see the double bonds (=) appear in different locations on each, as does the charge. Are they all the same molecule? Yes. Any way is an appropriate way to draw sulfate (even more are possible, just not as stable). More correctly than saying they are each correct, is to say that they all together are correct. Each depiction of the molecule is not real, the actual molecule is more like a combination of the 6. The double bonds do not really exist; rather the electrons are shared equally among all the bonds in this molecule, with only a probability of being “found” in any particular place. My o-chem instructor might say that the oxygens all have "1/2 a pie bond and a sigma." To see an organic molecule with even more resonance possibilities, check out benzaldehyde.
Which brings me to the next eerie point: the existence of electrons is only a probability, never an actuality. The orbitals we recall from high school chemistry are like the orbitals of the earth around the sun. This is not an accurate representation. Rather, orbitals are the most probable place to "find" an electron.
Below is a graph of the electron density probability of a simple s orbital:

The Y axis is the nucleus of the atom, the only place where the probability of finding the electron is zero. The density is most probable where we would normally draw the shell, all around the nucleus. This can also be represented by a “cloud” graph:

One thing to recognize about the electron density graph is that it progresses asymptotically; it never reaches a probability of zero as the electron travels away from the nucleus. Could it be that an electron from a hydrogen atom at the tip of my nose is somewhere flying out by the sun? Yes, just far less probable than the more likely ~5X10^-11 meters away from the nucleus of that atom.
Even stranger still, is that you’ll never run into an electron, whether it’s by the sun or on my nose. Electrons exist as waves (standing waves), not particles. So not only are they constantly moving, but they are defined as a movement. Even the deadest creature or mundane rock is very “lively” when we look at the atomic level. Electrons are restless and even neutrons within the core move about (that is how we get isotopes like C14 to date organic material). It turns out if you look close enough, every inch of our world is a very dynamic, ever changing place.
Links:
Check out my o-chem professor's page: http://oxygen.chem.uidaho.edu/davico/
And my bio-chem professor's: http://www.ag.uidaho.edu/mmbb/p_cole_d.htm
Sources:
Links:
Cytoskeletons: http://hsc.unm.edu/som/Research/
benzaldehyde: http://chemistry2.csudh.edu/rpendarvis/benzalNitrInt.GIF
Images:
Horse: http://www.powerhorse.com/photography/
Electron density probability graph and electron density cloud: http://www.physics.utoledo.edu/~adc/Chap-28_files/
Microfilaments: http://hsc.unm.edu/som/Research/technology.cfm
Sulfate resonance structures: http://www.chem.queensu.ca/people/faculty/mombourquette/FirstYrChem/Molecular/Lewis/index.htm
Sulfate: http://www.madsci.org/posts/archives/jan2000/


2 Comments:
amazing! i'm reminded of star wars for some reason--- May the electrons be with you! Or something like that.
By Anonymous, at 9:56 PM
Anonymous, at 9:56 PM
It actually makes more sense to me to define electrons as waves; makes me wonder less about them being little balls split into smaller and smaller particles and more about their nature in relation to the quintessence of energy and the purest form thereof.
I want to know more, but I figure by the time I get to that science class my head will already be full.
By Anonymous, at 2:38 PM
Anonymous, at 2:38 PM
Post a Comment
<< Home