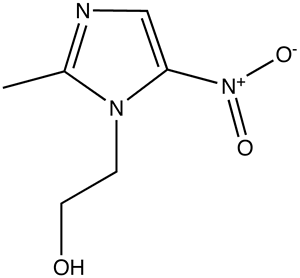
Recently I was placed on the antibiotic drug metronidazole (top) to treat an infection. In anaerobic (oxygen free) conditions, metronidazole acts on DNA repair enzymes and is an excellent tool against anaerobic bacteria such as Bacteroides, Clostridium, and Eubacterium while it does not harm the aerobic tissues of the human body.
Unfortunately, no alcoholic beverages can be consumed while taking this drug. Being a party-going college student, I became curious as to why I had to avoid this traditional Friday night favorite.
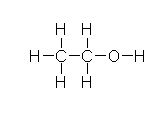 In the liver, alcohol (right) is broken down by alcohol dehydrogenases into acetaldehyde with the help of NAD by the following reaction: CH3CH2OH + NAD+ → CH3CHO + NADH + H+. Next, the acetaldehyde interacts with the acetaldehyde dehydrogenase (also in the liver) to form acetic acid (CH3-COOH, a very weak acid with many biological functions). Metronidazole is believed to obstruct the acetaldehyde dehydrogenase enzyme in much the same way disfulfiram, an anti-alcoholism drug, does. This allows excess acetaldehyde to build up in the body, 5-10 times as much acetaldehyde as one would normally experience after consuming an equal amount of alcohol with out the metronidazole. Since acetaldehyde is the primary cause of hangover, one can experience severe hangover-like symptoms including hot flash, shakes, nausea, and stomach cramps with even the slightest alcohol intake when combined with metronidazole.
In the liver, alcohol (right) is broken down by alcohol dehydrogenases into acetaldehyde with the help of NAD by the following reaction: CH3CH2OH + NAD+ → CH3CHO + NADH + H+. Next, the acetaldehyde interacts with the acetaldehyde dehydrogenase (also in the liver) to form acetic acid (CH3-COOH, a very weak acid with many biological functions). Metronidazole is believed to obstruct the acetaldehyde dehydrogenase enzyme in much the same way disfulfiram, an anti-alcoholism drug, does. This allows excess acetaldehyde to build up in the body, 5-10 times as much acetaldehyde as one would normally experience after consuming an equal amount of alcohol with out the metronidazole. Since acetaldehyde is the primary cause of hangover, one can experience severe hangover-like symptoms including hot flash, shakes, nausea, and stomach cramps with even the slightest alcohol intake when combined with metronidazole.Sources: Tillonen et al. Alcohol Clin Exp Res. “Metronidazole increases intracolonic but not peripheral blood acetaldehyde in chronic ethanol-treated rats.” 2000
http://www.healthyplace.com/medications/disulfiram.htm


0 Comments:
Post a Comment
<< Home