
Wheat: It's What's for Breakfast
Sponge cake. Spaghetti. Coco puffs. French loaf. All of these foods are certainly very distinct from one another (I know I’ve got to be in a particular mood for any one of 'em) yet they are all primarily a product of wheat. How does something so simple and seemingly homogenous as wheat come to make such vastly different foods?
Well, that's why wheat is only seemingly homogenous. Each strain of wheat has its own unique characteristics, much like the different breeds of dog. There are hundreds of kinds of wheat and wheat breeders are constantly trying to find a better wheat for the types of foods they're interested in producing.
It can be frustrating to know which strains to breed and running a trial all the way through to the market is quite costly when you consider the amount of energy gone into milling and processing. This is where my friend, food scientist Kameron Pecka, comes in. Kam works for the USDA Western Wheat Quality lab in Pullman, WA, where he, to put it bluntly, measures wheat hardness.
Measuring "hardness" is more complicated than it seems.
 First, Kameron must photograph the rectangularized wheat endosperm and take its dimensions. Then, using a TA-XT2 (photograph at right), Kam measures the stress and stain on the wheat. Next comes the number crunching: Kam uses graphs and equations involving the stress and stain to determine Young’s modulus, which indicates the elasticity of the wheat, and the total failure energy and force. All these figures come together to give an indication of hardness.
First, Kameron must photograph the rectangularized wheat endosperm and take its dimensions. Then, using a TA-XT2 (photograph at right), Kam measures the stress and stain on the wheat. Next comes the number crunching: Kam uses graphs and equations involving the stress and stain to determine Young’s modulus, which indicates the elasticity of the wheat, and the total failure energy and force. All these figures come together to give an indication of hardness."My work is more physics than biology," says Kam, who’s found himself faced with a calculator most days.
Putting Kam through all these calculations is not just an instrument of undergrad torture, though. Hardness is an indicator of many things that are extremely useful to wheat breeders. The texture of the wheat gives clues about its nutritional content, how much work must be put into milling, and what sort of foods it should be used for. Soft wheat typically goes into cookies, hard into certain breads, and durum is used to make pasta.
Currently Kam is busily working on a paper that will hopefully begin peer review in October. He’s excited by the possibility of being an undergraduate co-author.
As for "cracking wheat all day," Kam says his job is "monotonous, yet frighteningly satisfying."

The Drink!: Kam is enjoying a Hefeweizen, a German wheat beer which has not had the yeast filtered out. It’s extremely tasty when served with a slice of lemon.
Image sources: http://www.cals.ncsu.edu:8050/foodrheology/equipment/equipment.html
http://www.greenpeace.org/international/photosvideos/photos/wheat

 An excellent source of magnesium is raw, green vegetables as magnesium is central to each chlorophyll molecule. Chlorophyll are the powerhouse of plants, converting sun light to energy through and electron transport chain. They are what give plants their color as they absorb red light (680 and 700nm wavelengths) and reflect green. Some beans, nuts, fish, and whole grains are also an excellent source. More refined grains (such as in white bread) or cooked vegetables contain less magnesium than they would without processing. Tap water can even be a source of magnesium, if it is so called "hard water," which contains more minerals.
An excellent source of magnesium is raw, green vegetables as magnesium is central to each chlorophyll molecule. Chlorophyll are the powerhouse of plants, converting sun light to energy through and electron transport chain. They are what give plants their color as they absorb red light (680 and 700nm wavelengths) and reflect green. Some beans, nuts, fish, and whole grains are also an excellent source. More refined grains (such as in white bread) or cooked vegetables contain less magnesium than they would without processing. Tap water can even be a source of magnesium, if it is so called "hard water," which contains more minerals. Unfortunately, when stressed, these nitric oxide synthase enzymes can begin to produce excess NO and its oxidant product peroxynitrite (ONOO) leading to a chronic, cyclic increase in the quantities of both chemical products. The up-regulation of nitric oxide and peroxynitrite make up part of the vicious cycle Dr. Pall calls the "No!, Oh no!" cycle after the convenient way the chemical abbreviations for nitric oxide and peroxynitrite (NO/ONOO) can be read.
Unfortunately, when stressed, these nitric oxide synthase enzymes can begin to produce excess NO and its oxidant product peroxynitrite (ONOO) leading to a chronic, cyclic increase in the quantities of both chemical products. The up-regulation of nitric oxide and peroxynitrite make up part of the vicious cycle Dr. Pall calls the "No!, Oh no!" cycle after the convenient way the chemical abbreviations for nitric oxide and peroxynitrite (NO/ONOO) can be read.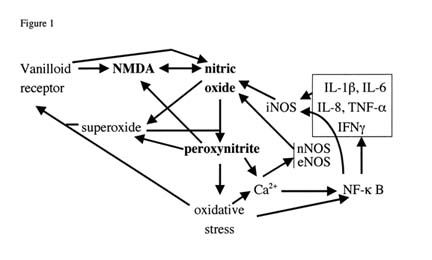

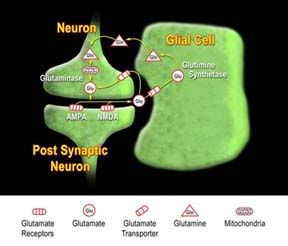







 Computers are able to perform more human-like function all the time as technology improves. Although I think (or is it hope?) Chobits and Blade-Runner are a long way off; surprising new technologies continue to appear with great frequency.
Computers are able to perform more human-like function all the time as technology improves. Although I think (or is it hope?) Chobits and Blade-Runner are a long way off; surprising new technologies continue to appear with great frequency.
 The Dark Side of the Potato
The Dark Side of the Potato

 The Socialization and Genetic Expression of Aggression in Danio Rerio (zebrafish)
The Socialization and Genetic Expression of Aggression in Danio Rerio (zebrafish)

